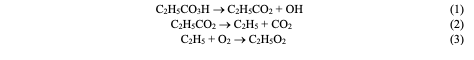|
Introduction The possibility, of heterogeneous interactions between CH3O2 radicals with aldehydes and hydrocarbons has been established in recent studies [1−9]. However, there is no direct evidence of such heterogeneous reactions concerning C2H5O2 and C2H5 radicals. In this study, we sought to provide such evidence by analyzing and discussing the kinetic data obtained from the investigation of the influence of the reaction vessel surface (H3BO3, KCl) on the oxidation process of propionaldehyde at atmospheric pressure in air flow [10,11]. This reaction proceeds via a chain degenerate branching mechanism [12].
Eq. 1-3, where the branching stage at rela-tively low temperatures, involves the heterogeneous radical decay of C2H5CO3H (Eq. 1).The leading active centers in this process are C2H5CO3 radicals (Eq. 1-3). As the oxidation process develops, the contribution of C2H5O2 radicals, formed as a result of the heterogeneous decay of peracid, increases(Eq. 1). Based on the dependence of the yields ofsuch reaction products as ethylene and ethyl hydroperoxide on the nature of the reactor surface [10,11], it was possible to identify their heterogeneous formation with the participation of C2H5 radicals (Eq. 2, 3). Gas-phase oxidation reactions of aldehydes,proceeding via a chain degenerate branching mechanism, are a source of valuable oxygen-containing compounds. Depending on the nature of the reaction vessel, both the rate and the direction of the process can change [12]. For example, during the oxidation of C2H5CHO in the boric acid-treated reactor, peroxypropionic acid (C2H5CO3H) can be formed with high selectivity (80%),while in the potassium chloride-treated reactor, the selectivity for propionic acid (C2H5CO2H) is 90%. The heterogeneous reactions of peroxy radicals such as CH3O2 play a crucial role in thecombustion of CH3CHO + O2 mixtures,initiated by peroxy radicals formed during the heterogeneous radical decay of RCO3H[13]. Thus, investigating the possibility ofheterogeneous reactions involving C2H5O2 (Eq. 3) and C2H5 (Eq. 2) radicals is essential for understanding the mechanism ofoxidation and combustion reactions of aldehydes.
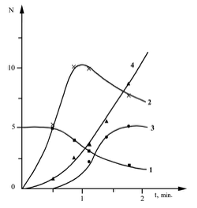
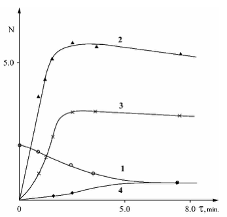
Results and Discussion Reaction of C2H5O2 Radical with Aldehyde on Solid Surface Figures 1 and 2 demonstrate the kinetic curves of the consumption of C2H5CHO and the accumulation of some reaction products including peroxy radicals, C2H5CO3H and C2H5O2H, obtained in the aforementioned two reactors at 291oC [11].
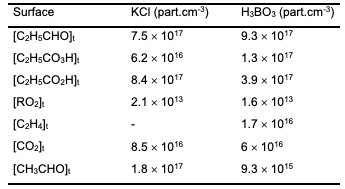
Table 1 shows the concentrations of reaction products at 291°C and 1 minute of reaction time in reactors treated by boric acid and potassium chloride.
This data indicates that the process characteristics strongly depend on the nature of the surface. This applies both to the composition of the reaction products and to the absolute values of products concentrations. Notably, C2H5CO3H is absent in the reactortreated with KCl. This is attributed to a higher rate of heterogeneous radical decay of the peracid on the KCl surface in compared to the H3BO3 surface [14]. The high yield of CO2, formed during the decayof peracid in the reactor, further supports this. Consequently, the process in the KCl-treated reactor begins earlier than in the H3BO3-treated reactor, and even at t = 1 min the aldehyde consumption is 1.4 times greater than that in a boric acid-treated reactor. It is noteworthy that the concentration of peroxy radicals in the volume is less by approximately 2 times. This clearly indicates that a significant portion of the aldehyde in the KCl-treated reactor is consumed on the reactor surface. The yieldof hydroperoxide is almost three times higher than in the reactor treated with boricacid. Apparently, a significant contribution to the formation of hydroperoxide is linked to the heterogeneous interaction of C2H5O2 radicals with propionaldehyde. The presence of ethylene in the KCl-treated reactor and its absence in the boric-acid reactor, also suggests that more C2H5 radicals were formed in the first reactor. It is important to note that the reactions of C2H5 radicals are the source of both ethylene and C2H5O2 radicals. Regarding the H3BO3 surface, since it is known that the rate of C2H5CO3H heterogeneous radical decomposition on this surface is much slower than that on the KCl surface, it is evident that the amount of formed C2H5O2 radicals on the H3BO3 surface should be significantly lower. Therefore, the yield of hydroperoxide is also lower. A comparison of the kinetic curves for the accumulation of peroxy radicals in both reactors and, considering the significant amounts of C2H5CO3H in the boric acid-treated reactor, shows that the ratio of C2H5CO3 and C2H5O2 radical concentrations is higher in the boric acid-treated reactor. Analysis of the kinetic data on propionalde-hyde oxidation depending on the nature of the surface, reliably indicates the possibilityof heterogeneous interaction between C2H5O2 radicals and aldehyde-C2H5CHO.Thus, we conclude that the interaction ofperoxy radicals with organic compounds ismore general and is not limited to CH3O2radicals.
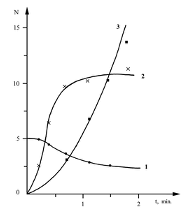
Heterogeneous Reaction Pathways of C2H5 Radicals on a Solid Surface This work presents data on the heterogeneous formation of several reaction products, such as C2H4 and C2H5O2H, during the low-temperature oxidation of propionaldehyde. These results were obtained by analyzing the kinetic patterns of the process in reactors treated with boric acid and potassium chloride [10]. Fig. 3 and 4 illustrate the kinetics of peroxyradicals formation and the initial aldehyde consumption while the concentrations of some reaction products for a fixed reaction time are shown in the Table 2.
Comparison of the results obtained in thetwo reactors reveals that the rate of the process in the potassium chloride-treated reactor is higher than in the boric acid-treated reactor, while the yield of C2H5CO3H product responsible for the branching is much lower. This was not unexpected, as the rate of heterogeneous radical decay of peracids is greater in a reactor treated with potassium chloride [14]. The data indicates that the concentration ofperoxy radicals such as C2H5CO3 and C2H5O2 [10] in the given volume, correlates with the rate of oxidation in these reactors,and is higher in the reactor treated bypotassium chloride. An important feature of the process is the detection of ethylene and the absence of hydroperoxide in the boric acid-treated reactor. Conversely, in the potassium chloride-treated reactor, hydroperoxide is detected while ethylene is absent. This suggests that there are heterogeneous pathways for the formation of these compounds. Possible pathways for the formation of these compounds are: It is noteworthy that comparison of the yields of ethylene and hydroperoxide with those obtained during the oxidation of C2H5CHO in the same reactors at 291oC [11], reveals that the concentration of hydroperoxide is higher in the reactor treated with potassium chloride, although a certain amount of it is also already registered in the reactor treated with boric acid. As for ethylene, it is formed at higher temperatures in the potassium chloride-treated reactor. The obtained data show that the competition between reactions 2. and reactions 4. and 5. favor of 2. on the KCl surface.
Conclusion Thus, this study allows us to conclude that the heterogeneous reactions of C2H5O2 and C2H5 radicals play a crucial role in the gas-phase oxidation of C2H5CHO, contributing to the formation of several reaction products. Declaration of competing interest: Theauthors declare that they have no knowncompeting financial interests or personalrelationships that could have appeared toinfluence the work reported in this paper.
References
|
||||||||||||||||||||||||||||||