|
Introduction The issue of corrosion of steel reinforcement and structural steel is considered one of the most important problems facing several industries, such as construction, marine, and oil industries [1,2]. Solving this issue entails large maintenance expenditures because it creates a major threat to the safety of facilities. Engineers used several solutions to tackle this issue including cathodic protection [3], concrete permeability reduction to block corrosion agents [4], and applying anticorrosion coatings to metal surfaces [5]. Because of the significance of the corrosion problem, there have been mathematical models that predict it. For instance, Asmara and Kurniawan [6] researched the predictive models and methods of corrosion rate for carbon steel, which is a major problem in oil and gas. They also examined the environmental conditions affecting corrosion, i.e., temperature, pressure, and chemical composition, while assessing predictive models derived from experimental data and computer simulation in their research. Other researchers [7] addressed the topic of interest to most industrial processes, i.e., corrosion in mild steel when exposed to acidic environments. The researchers investigated the possibility of using readily available eco-friendly imidazolium-based ionic liquids as corrosion inhibitors. They employed both experimental techniques and theoretical models to explore inhibition mechanisms. The results show that these ionic liquids are efficient in inhibiting corrosion and that some molecular interactions are important in the process. Similarly, how metals corrode in different industry environments was explored [8]. The corrosion resistance of AISI304 stainless steel and copper when immersed in alkali (KOH and NaOH) and saline (NaCl) solutions under the electrolysis process was the subject of the experiment. They performed electrochemical tests to determine the corrosion rate of this alloy and found that it was more prone to corrosion when immersed in NaCl because of the aggressive chloride ions. Conversely, the rates of corrosion of KOH and NaOH were lower, but KOH offered more protection to the stainless steel. The authors underlined the need to select material for the intended purpose in a given industrial environment. Coatings are an intricate multi-step process rooted in basic chemistry and physics to coat metal surfaces against corrosion. In its simplest mani- festation, this process is designed to create a barrier that prevents corrosive substances like chlorine, oxygen, and water ions from making contact with the metal surface and triggering destructive interactions [9,10]. Coatings like these work partly because they mechanically block electrons from the metal surface, but they also establish a barrier to the transfer of charge in general. This keeps electronic oxidation, the first stage of corrosion, from happening [11]. Through the combination of these two principles, anti-corrosion coatings can strengthen the corrosion barrier in a way that neither of them could do alone. The coating protects the corrosive chemical substances from the exposed metal surface by forming a physical barrier. At the same time, corrosion-causing electron flow is blocked by its high electrical resistance. On the surface of the metal, the coating effectively inhibits electronic oxidation reactions from starting by blocking charge transfer. The discovery of nanomaterials and their use to improve physical and chemical properties of materials revolutionized the field of materials science since nanomaterials have more superior mechanical properties com- pared to their bulk counterparts. For instance, carbon nanotubes (CNTs) and graphene have superior strength, stiffness, and flexibility [12], which make them able to reinforce polymers, ceramics, and metals in composites. This results in materials of higher hardness and strength and resistance to corrosion and fatigue. At the chemical properties level, nanomaterials have a high surface-to-volume ratio, which provides more surface area for interaction with their surrounding environments [13]. It should be remembered that nanomaterials have played a very important role in improving the physical properties of many coatings, for example, improving thermal conductivity and optical absorption [14] in solar collector coatings, resistance to ultraviolet radiation [15], fungi and bacteria resistance [16], and corrosion resistance [17]. In this regard, collective research endeavors have been made to explore the intricate dynamics among various nano-additives and how they enhance the anti-corrosion properties of the coating. A core research interest in this regard is nanoparticles (NPs) developed from metals or their oxides. These NPs have been comprehensively studied to determine their role in enhancing the anti-corrosion properties of coatings [18]. Also, the work includes nanoparticles from carbon-based materials, i.e., graphene and carbon nanotubes, which are greatly prized for their excellent mechanical, electrical, and chemical properties. The work has included both these pure carbon materials and their oxides, i.e., graphene oxide. Following our earlier work [18], we investigated the influence of adding metal or metal oxide nanoparticles on the anti-corrosion behavior of coatings. This work intends to compare the impact of integrating diverse carbon-based NPs, namely graphene, graphene oxide, and carbon nanotubes, on the anti-corrosion capabilities of polymeric coatings. We aim to determine the most effective nano-additive for increasing corrosion resistance by assessing its impact on barrier characteristics, mechanical reinforcement, and overall protective efficacy. The results will yield remarkable insights into the ideal selection of nano-fillers for the formulation of sophisticated anti-corrosion coatings.
Experimental In our study, a comparative analytical methodology was used to examine the results obtained from previous studies conducted by researchers in the field of anti-corrosion coatings research, specifically those involving the effect of carbon nanomaterials on anti-corrosion properties. In order to compare the protection effectiveness resulting from coatings modified with different carbon nanomaterials, the protection effectiveness provided by these coatings was calculated based on the corrosion current values before and after applying the modified coating (these values are mentioned in the studies we discussed in this review or in the supplementary materials attached to those studies).
Results and Discussion The incorporation of carbon NPs into polymeric coatings has demonstrated huge potential in the enhancement of corrosion resistance, which leverages the distinctive physical and chemical properties of materials such as graphene, carbon nanotubes (CNTs), and graphene oxide (GO). Graphene, haracterized by high electrical conductivity, exceptional mechanical strength, and a large surface area, enhances corrosion resistance through its impermeable nature and effectively blocks the diffusion of corrosive agents like water and oxygen. Similarly, CNTs improve anti-corrosion properties by forming a reinforcing network within the polymer matrix due to their outstanding tensile strength and flexibility. Thereby, the structural integrity and hindering the movement of corrosive substances are increased. Overall, it can be stated that carbon-based NPs introduce a more tortuous path for corrosive agents that reduce their penetration rate and modify the electrochemical properties of the coating to enhance corrosion resistance. Additionally, the mechanical properties of the coating are improved, which gives more resilience to physical damage that could expose the underlying metal. The following sections explore the materials mentioned above.
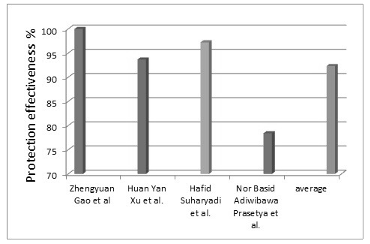
Graphene Graphene-modified anti-corrosion coatings have emerged as a highly effective solution to enhance the durability and longevity of materials exposed to corrosive environments. Incorporating graphene into polymeric coatings enhances its physical and chemical properties; this leads to noticeable improvements in wear resistance. The incorporation of graphene into the polymeric coating matrix increases its corrosion resistance through two mechanisms. The first mechanism is termed the barrier effect, which is one of the strongest mechanisms for corrosion protection. On this basis, the impermeable nature of graphene prevents the diffusion of corrosion-causing agents such as water, oxygen, and salts. This decreases the rate with which these agents seep down to the supporting mineral substrate markedly. The other is mechanical strengthening, where the polymer matrix receives a reinforcement through the addition of graphene to augment its mechanical stiffness. Here, the possibility for cracks and faults that expose metal to corrosive substances is curbed. In another study by Gao et al. [19], an oil-based epoxy resin (OEP) coating modified with graphene (G/OEP) was synthesized to enhance corrosion resistance of AZ31 magnesium alloy. In this study, the potential benefits of using graphene in epoxy resin coatings in surface finish, structure, and protection were taken into consideration. AZ31 magnesium alloy was coated with G/OEP, i.e., graphene-modified oil-based epoxy resin, for this study. For comparison of the coatings that had been modified using graphene with the unmodified coatings, as well as for detecting surface defects, scanning electron microscopy (SEM) observations were also conducted. Surface finish of the coating and defect reduction were both enhanced by the addition of graphene, as revealed through SEM micrographs. FTIR spectroscopy analysis of the coating composition ensured that the coatings were primarily composed of epoxy (polyurethane) resin and its curing agent, with the addition of graphene incorporated in the matrix successfully. The corrosion resistance of the coatings was also assessed using electrochemical measurements. The results indicated that the G/OEP coatings provide excellent corrosion protection, as the corrosion current density of the magnesium alloy reduced from 6.20 × 10−7 A/cm2 (0 wt.% graphene) to 6.96 × 10−12 A/cm2 (0.6 wt.% graphene) upon the application of the G /OEP coatings, which increased the protective efficiency to 99.99%. Finally, a post-corrosion morphology analysis was performed to examine the structural integrity of the coating after being subjected to electrochemical corrosion. The analysis aimed to assess the extent of structural damage and determine the role of graphene in enhancing the protective nature of the coating against corrosive media. The results indicated that graphene significantly improved the corrosion resistance of the coating by reducing structural damage. The incorporation of graphene effectively sealed micro-defects in the coating upon curing, hence inhibiting the uptake of corrosive materials and enhancing the durability and protective efficacy of the coating as a whole. In a study by Xu et al. [20] for enhanced anticorrosion properties of composite coatings, E44 waterborne epoxy resin was used as the matrix material, and graphene was incorporated as a nano-filler. The investigation focused on studying the structure and electrochemical response of such composite coatings with a view to studying the contribution of graphene toward increased corrosion resistance. To characterize the graphene, X-ray diffraction (XRD) and transmission electron microscopy (TEM) were used. The resulting structure is a thin pile of graphite sheets with many wrinkles, and it improves the coating performance. Graphene-epoxy coatings were found to have a flawless surface according to SEM. This was attributed to the ability of the graphene to effectively fill micro-pores and internal micro-cracks, thus strengthening the coating and making it more protective. The FTIR test results indicated that the composite coatings were primarily composed of epoxy resins with polyamide curing, in which the characteristic peaks according to the materials used. Finally, the result of electrochemical testing, particularly the Tafel polarization curves, indicated that it raised the corrosion potential and reduced the corrosion current when more graphene was incorporated. Therefore, the corrosion resistance was ultimately enhanced. The intensity of the corrosion current decreased from 1.2 × 10–3 A·cm–2 in the case of pure epoxy to 7.6 × 10–5 A·cm−2 in the case of epoxy with 0.6 wt.% nano-filler, which means an increase in the effectiveness of protection by 93.67%. However, the electrochemical impedance spectroscopy (EIS) results showed that graphene increased the impedance arc in Nyquist plots and impedance modulus at low frequencies, confirming the improvement in corrosion resistance. The study concluded that the improved mechanism of corrosion protection by graphene is attributed to the improved barrier performance of the coating because graphene clogs the pores in the coating matrix. Thus, penetration of the corrosive substance was avoided, and the general long-term stability of the coating was improved. Suharyadi et al. [21] studied the effect of graphene and epoxy coatings as corrosion inhibitors on carbon steel plate substrates used in heat exchangers in the oil and gas industries. Several coating samples were prepared with different mass concentrations of graphene/epoxy (0%, 2%, 5%, and 10%), and the coating was applied to the substrates using the bath method. The corrosion behaviors of substrates immersed in 1 M sulfuric acid solution at room temperature were studied using dynamic polarization. Experimental results indicated that graphene and epoxy coatings are effective in inhibiting corrosion. The dynamic polarization potential curves showed that the corrosion potential of coated carbon steel turned to more positive values than that of uncoated carbon steel. The study also found that corrosion current densities and corrosion rates decreased by up to 36 times. This improvement was attributed to the enhanced barrier effect of the coating, achieved by filling its pores with well-dispersed graphene nanoparticles. This modification reduced the permeability of the graphene-epoxy coating to ions and other corrosive species in acidic solutions, ultimately increasing its protective effectiveness to 97.23%. In another study [22], an anticorrosion coating composed of a polyeugenol/graphene (PE/G) composite was developed, and its effectiveness in protecting metals from corrosion was evaluated. Here, several methodologies were used to synthesize and evaluate the PE/G composite. Polyeugenol was synthesized via cationic addition polymerization using BF3O(C2H5)2 as a catalyst, while graphene was produced through the chemical reduction of GO using the Hummers' method. The PE/G composite was prepared at various weight concen- trations (0.25 wt.%, 0.5 wt.%, 0.75 wt.%, 1 wt. %, and 1.25 wt.%.) using a solution mixing method and applied to metal substrates via drop-casting. To assess the anticorrosion performance, the researchers used potentiodynamic polarization techni- ques, which involved measuring the electro- chemical behavior of the coated metals in a 3.5% sodium chloride solution. The PE/G composite exhibited superior corrosion resis- tance compared to unmodified polyeugenol, as the addition of graphene reduced the corrosion current density from 4.0239 µA·cm−2 at 0 wt.% to 1.3854 µA·cm−2 at 1.25 wt.% of graphene, leading to a protection efficiency increase of up to 78.42%. Morphological analyses were conducted using SEM which revealed that the PE/G coated metals had a defect-free surface and the graphene was effectively filled within the micro-pores and micro-cracks. XRD analysis was also performed, and the obtained results confirmed the absence of corrosion products on the PE/G-coated metals after exposure to the corrosive environment. Accordingly, the studies reviewed clearly show that graphene has a great ability to improve corrosion-resistant properties, as the average protection effectiveness is about 96.963%.
Figure 1 shows the values of the corrosion protection effectiveness of graphene-con- taining coatings and the average value of selected studies.
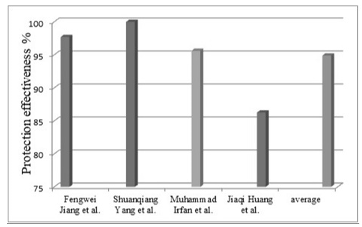
Graphene Oxide (GO) Due to the unique properties of graphene oxide (GO), such as high surface area, excellent mechanical strength, and excellent barrier properties, it has been one of the most promising additives to enhance the performance of corrosion-resistant coatings. GO is a graphene derivative that contains oxygen functional groups, such as hydroxyl, epoxide, carbonyl, and carboxyl groups, which enhances its dispersibility in water and organic solvents. These functional groups also allow GO to form strong interactions with polymer matrices and make it an excellent candidate for composite coatings. The high aspect ratio of GO creates tortuous paths that impede the diffusion of corrosive agents, greatly improving the barrier pro- perties of the coating. Regarding corrosion resistance mechanisms, the effectiveness of GO in corrosion-resistant coatings can be attributed to several mechanisms. The first one is the barrier effect, where the layered structure of GO increases the diffusion of corrosive species. Here, a delay in the penetration of water, oxygen, and ions occurs. The second mechanism is its self-healing properties, as some studies [23-25] indicate that combining GO with some polymers can contribute to repairing small cracks independently and prolong the life of the coating. The third is the synergistic effects with polymers, where GO can interact with polymer matrices to enhance their mechanical properties and adhesion to substrates, and the coating failure is possibly reduced. In their study, Jiang et al. [26] investigated the corrosion resistance behaviors of epoxy composite coatings reinforced with GO. They focused on understanding how varying aspect ratios of GO influence the corrosion resistance properties of the coatings. To achieve this, the researchers prepared epoxy composite coatings by incorporating GO with different aspect ratios. The study sought to understand better how GO's structural properties affect its ability to improve composite coatings' anti-corrosion performance. As a result, valuable insights that can be used to optimize the design of advanced protective coatings were gained. The researchers used a modified Hammers method to synthesize GO in different aspect ratios. They then mixed it with an epoxy resin matrix at a concentration of 0.1 weight percent. The composite coatings were applied to sandblasted carbon steel metal substrates. To evaluate the corrosion resistance performance, a series of electrochemical tests were performed, including dynamic polarization and EIS, the results of which indicated that the aspect ratio of GO highly affected the corrosion resistance of epoxy composite coatings, where coatings with a higher aspect ratio of GO had superior corrosion resistance. The corrosion current densities decreased significantly from 3.115 × 10-7 A·cm-2 for GO-c to 1.164 × 10-8 A·cm-2 for GO-a, which demonstrates a substantial improvement in protection effectiveness of approximately 97.71% compared to the neat epoxy coating. Concurrently, the complex impedance values increased from 2 × 106 to 4 × 107, further confirming the enhanced barrier properties and corrosion resistance of the GO- reinforced epoxy composite coatings. In addition, the surface morphology and structural properties of the coatings were examined using SEM and TEM. These analyses determined that there was a rise in the uniformity and defect-free surface of the coating as the GO aspect ratio was greater. This increased structural integrity signifi- cantly contributed to the barrier nature of the coatings, which ensured that the corrosive agents were not able to penetrate and added further to the overall corrosion resistance of the composite coating. The study concluded that increasing the aspect ratio of GO in epoxy composites is vital in order to make the corrosion resistance more efficient. In another research conducted by Yang et al. [27], the in situ polymerization method was used for preparing GO/polyaniline (GO/PANI) nanocomposites with unique anti-corrosion properties. In this research, epoxy/GO coating samples were prepared. In order to characterize the different samples, surface morphology of the materials was studied using SEM, whose images revealed that pure GO had flaky structure with a folded and smooth surface. For understanding corrosion resistance, EIS measurements and dynamic polarization curves were carried out. The findings of these tests indicated that the incorporation of graphene caused Ecorr to rise from −0.704 volts for the neat epoxy to −0.548 volts for the epoxy/GO. Additionally, corrosion current density (Icorr) was decreased from 3.64 × 10-4 A/cm-2 for pure epoxy to 1.79 × 10-8 A/cm-2 for epoxy/GO, which can be an indication of 99.99% improvement in protection efficiency. In another study, Irfan et al. [28] explored the performance of a new anti-corrosion and environmentally friendly coating was developed and evaluated. The study focused on the utilization of a hybrid nanocomposite coating that includes composite green NPs and biopolymers to enhance corrosion resistance on metal substrates. The methodology involved the NPs synthesis process using plant extracts as reducing agents, and then nano-hybrid coatings were prepared by dispersing the composite NPs in a biopolymer matrix. Electrochemical methods, including dynamic polarization and EIS, evaluated the corrosion resistance of the coatings. Experimental results showed that nano-hybrid coatings improved the corrosion resistance of metal substrates, where electrochemical tests revealed that the coatings effectively reduced the Icorr, as its value decreased from 1.767 × 10-7 A·cm-2 for the coating without nano-filler to 7.736 × 10-9 A·cm-2 for the coating with nano-filler, which means the effectiveness of protecting the coating against corrosion increased by 95.62%, and the impedance value (Ecorr) increased from -0.476 volts for the paint without nano-filler to -0.191 volts for the coating with the NPs filler, indicating enhanced protective properties. SEM and TEM analyses showed that the NPs were well dispersed within the polymer matrix, which contributed to a uniform and defect-free coating surface. In a study by Huang et al. [29], waterborne epoxy coatings with enhanced wear resistance were developed by incorporating NPs of aluminum oxide (Al2O3), GO, and hybrid Al2O3@GO. In order to prepare composite coatings, the modified NPs were dispersed in waterborne epoxy resin, where different concentrations of these NPs (1.5 wt.% for Al2O3, 0.2 wt.% for GO, and 0.4 wt.% for Al2O3@GO) were used to create different composite layers. Then, to characterize the samples, the researchers used several techniques to characterize the surface morphology and the morphology of NPs. The SEM images showed the presence of ripples in a large area of the GO sheets, which represents a stacked state, where the GO sheets were strongly assembled into a stacked shape with a width of micrometers. Regarding corrosion resistance tests, dynamic polarization tests and electro-chemical impedance spectroscopy were conducted to evaluate the coatings in a 3.5% sodium chloride solution. The results showed that coatings that contain GO showed superior corrosion resistance compared to neat epoxy coatings. The value of the corrosion potential Ecorr increased from −0.760 V in the case of neat epoxy to −0.354 V in the case of 0.4% epoxy/GO coating. Also, the value of the Icorr decreased from 1.235 × 10-5 A·cm-2 in the case of neat epoxy to 1.697 × 10−6 A·cm-2 in the case of 0.4% epoxy/GO coating. These results indicate an increase in protection effectiveness by 86.26%. The improved performance is attributed to the better barrier properties and lower permeability provided by the well- dispersed NPs. Accordingly, the studies reviewed clearly show that GO has a great ability to improve corrosion-resistant properties, as the average protection effectiveness is about 94.895%. Figure 2 below shows the values of the corrosion protection effectiveness of GO- containing coatings and the average value of selected studies.
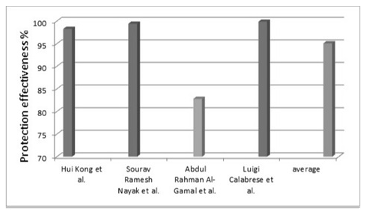
Carbon Nanotubes Carbon nanotubes (CNTs) show promise as an additive in anti-corrosion paints due to their high mechanical properties due to their high tensile strength and better chemical properties like chemical inertness. These features make CNTs suitable candidates to enhance the performance of anti-corrosion coatings. CNTs aid corrosion protection by multiple mechanisms, for which the most important one is the barrier effect. CNTs create a dense and impermeable network within the coating matrix, which increases the diffusion of corrosive species such as water, oxygen, and chloride ions to the substrate. The mechanical strengthening mechanism is another critical parameter in which CNTs enhance the corrosion resistance of the coating. By enhancing the mechanical properties of the coating, CNTs become physically more robust and reduce the risk of exposing the metal substrate to corrosive conditions. In a study by Kong et al. [30], a corrosion-resistant polymeric coating composed of polyurethane and CNTs was prepared. The study was focused on the effectiveness of using NPs in improving the mechanical and anti-corrosion properties of such coatings. In preparation of the coating, the researchers incorporated NPs into the polymeric matrix, with varying concen- trations of NPs used to find the optimal formulation for optimal performance. SEM analysis was performed to examine the surface morphology, and dispersion of NPs within the coatings. The NPs were homogenously dispersed in the images, adding to the global properties of the coating. They were uniformly distributed in the polymeric matrix and formed defect-free coatings. Corrosion protection of the coatings for electrochemical tests was analyzed using methods such as Tafel polarization and EIS. Electrochemical analysis showed that the addition of CNTs caused an increase in the Ecorr from −0.54 V for the plain coating to −0.38 volts for the 3% CNTs-containing paint. The value of the Icorr decreased from 5.24 × 10-7 A·cm-2 in the case of coating without nano-fillers to 8.94 × 10−9 A·cm-2 in the case of the coating of 3.0% of CNTs, which means an increase in protection effectiveness of 98.29%. In another study [31], a corrosion-resistant coating composed of epoxy coating and functionalized CNTs was developed and evaluated to enhance the corrosion resistance of aluminum alloys. The primary goal was to explore the effectiveness of incorporating these NPs into epoxy coatings to improve their mechanical properties and protect aluminum substrates from corrosive environments. To prepare the coating, the researchers incorporated functionalized CNTs into an epoxy matrix by dispersing the modified NPs in epoxy resin. The mechanical stirring and ultrasonic dispersion techniques were used after these NPs had been modified with appropriate chemical treatments to enhance their dispersion and compatibility with the epoxy resin. Finally, the resulting nanocoatings were applied to aluminum alloy substrates using spray coating processes. Also, SEM was used to characterize the surface morphology and ensure uniform dispersion of the NPs within the coatings. The SEM images showed that running the NPs led to their excellent dispersion within the epoxy matrix, as the nanocomposite coatings had a uniform surface free of defects. For electrochemical tests, techniques such as Tafel polarization and EIS have been used to evaluate the corrosion resistance of the coating. These tests were performed in a 3.5% sodium chloride solution to simulate a corrosive environment. Electrochemical tests indicated a significant improvement in the corrosion resistance of the nanocoatings compared to the unmodified epoxy coatings. The Tafel polarization curves showed a huge decrease in the corrosion current density, as its value decreased from 7.155 × 10-7 A·cm-2 in the case of the neat epoxy coating to 4.050 × 10-9 A·cm-2 in the case of the coating containing 0.25% CNTs, where the effectiveness of corrosion protection reached 99.43%. The EIS results also showed an increase in impedance, as the Ecorr value increased from −0.594 V in the case of the neat epoxy coating to −0.001 V in the case of the coating containing 0.25% CNTs, which indicates better barrier properties of the coatings. In a similar context, Al-Gamal et al. [32] developed a polymeric coating made of polyaniline (PANI) and alkyd resin containing multi-wall carbon nanotubes (MWCNTs) to enhance their corrosion resistance properties. To prepare the coating, the researchers chemically modified CNTs to improve their dispersion and compatibility with polyaniline resins. Then, different concentrations of functionalized CNTs were then incorporated into the PANI resin matrix. After this, coatings were prepared by mixing short alkyd resin as a binder with selective dyes for several pigment/binder mixing ratios, and then the solvent, which is xylene, was added at a rate of 15% by weight. Finally, the paint was applied to the carbon steel with a brush to prepare the samples. The SEM images confirmed the uniform distribution of CNTs in the coating matrix. Additionally, the corrosion resistance of the coating was characterized by electrochemical tests, in which Tafel polarization was performed. The results of this test indicated an improvement in the corrosion resistance of the coating containing the nanocomposite PANI/MWCNTs compared to the unmodified coating, where Tafel polarization curves showed a significant decrease in the Icoor, as its value decreased from 5.99 μA in the case of PANI/alkyd coating to 1.030 μA in the case of (PANI-MWCNTs)/alkyd 3% coating, which indicates enhanced protection against corrosion as it was protection effectiveness of 82.81%. A corrosion-resistant coating was prepared to protect aluminum surfaces [33]. The coating consisted of a sol-gel of N-propyl-trimethoxy-silane filled with different amounts of MWCNTs. The coating was applied to aluminum substrates by drop casting. The SEM images showed that a uniform gel coating with a content of 0.4 wt.% CNT was obtained. Low or high nanotube contents cause non-homogeneity of nano-filler distribution in the coating. The corrosion resistance using dynamic polarization and electrochemical impedance analysis when immersed in 3.5 wt.% sodium chloride was also characterized. The results of the analysis of dynamic polarization curves indicated a decrease in the Icoor, as its value decreased from 0.8 × 10-2 mA/cm-2 in the case of the coating without nano-fillers to 1.0 × 10-5 mA/cm-2 in the case of AS3-CNT6 nano-fillers, which means increased protection effectiveness by 99.875%. The electrochemical impedance analysis results indicated a decrease in the Ecorr value from - 1.210 volts for the paint without nano-fillers to −0.220 volts for the AS3-CNT6 nano- fillers. Accordingly, the studies reviewed clearly show that CNTs can improve the corrosion- resistant properties of coatings, as the average protection effectiveness value is about 95.10%.
Figure 3 shows the corrosion protection effectiveness of CNT-containing coatings and the average value. It is worth noting that the results of the studies showed that the modification of poly- meric coatings using CNTs provided the highest performance in reducing corrosion, followed by GO and, finally, graphene. The average improvement in protection effectiveness supports this finding. This can be attributed to the unique structure of carbon nanotubes (CNTs), which, as one- dimensional (1D) particles, can occupy spaces in the polymer matrix that two-dimensional (2D) graphene oxide (GO) or graphene particles cannot. As a result, CNTs exhibit a greater ability to enhance the barrier effect compared to GO or graphene particles. On the other hand, the dispersion of GO could be better than the dispersion of graphene in the polymer matrix. This can be attributed to several reasons, the most important of which is that GO contains different oxygen functional groups, such as hydroxyl and carboxyl groups, which enhance its interaction with polymer matrices. The absence of functional groups in graphene could also lead to large van der Waals forces between individual graphene sheets and cause aggregation in the polymer matrix. These forces are greatly weakened when graphene is oxidized, which improves its dispersion in the coating matrix. This, in turn, improves the barrier effect, which leads to improved protection effectiveness of the coating.
Conclusion This research presents a comprehensive review of the impact of incorporating carbon nanomaterials, specifically graphene, GO, and CNTs, as fillers within polymer-based corrosion-resistant coatings on the corrosion resistance properties of these coatings. Based on data from the relevant literature, the average protection effectiveness of each type of nanomaterial was determined. The analysis revealed that CNTs offered the highest corrosion reduction performance, followed by GO and graphene. The variation in protection effectiveness for the same type of additive can be attributed to several factors, primarily the type of polymer forming the host matrix. Differences in polymer composition influence its bonding with the nano-additive, while the polymer matrix density affects the amount of nano-filler that can be incorporated without causing agglomeration. Achieving a stable, homogeneous coating enhances its ability to form an effective barrier against corrosive agents. This study provides researchers with insights for selecting the most suitable type of nano-filler for the host coating matrix.
References
|