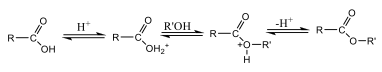|
Introduction Esterification is a foundational reaction in organic chemistry, producing esters, a versatile class of compounds widely used across industries. These carbonyl-containing molecules, both acyclic and cyclic, serve as essential building blocks in organic transformations, finding applications in fine chemicals, polymers, pharmaceuticals, cos- metics, and personal care products [1]. Esters have been integral to industrial chemistry since their first synthesis by German chemist Leopold Gmelin [2]. They remain one of the most abundant functional groups in both synthetic and natural compounds, emphasizing their central role in chemical synthesis and their extensive applications. Typically, esters are synthesized through the esterification reaction, wherein carboxylic acids and alcohols react in the presence of a catalyst. Other methods, such as reactions with carboxylic anhydrides or halides, are also employed for ester production [2]. This synthesis is increasingly significant due to esters' unique applications in various fields, including biodegradable polymers, thermoplastics, pharmaceuticals, agrochemicals, cosmetics, and food additives. For instance, esters like polylactic acid and polyethylene terephthalate are pivotal in producing bio- degradable plastics, while others add flavors to foods or fragrances to personal care products [3]. Additionally, ester derivatives, such as benzyl benzoate, methyl benzoate, and benzyl acetate, demonstrate therapeutic and antimicrobial properties, further highlighting their importance in pharmaceuticals [4]. This review aims to provide an in-depth analysis of current advancements and emerging trends in ester synthesis, with particular attention to new catalytic strategies and green synthesis approaches. These recent method- ologies address critical industrial needs for more efficient, sustainable, and application-specific ester production processes. By ex- amining these advancements, we highlight the innovative pathways that researchers are exploring to enhance ester synthesis, expand- ing its practical and commercial potential across multiple fields.
Synthesis of Esters Esters are structurally versatile compoundswith significant industrial relevance, synthesized primarily via esterification processes that vary in terms of reactivity, catalyst requirements, and environmentalimpact [5]. Traditional esterification methods involve the reaction of carboxylic acids with alcohols, acid anhydrides with alcohols, or acid chlorides with alcohols. Each route presents unique advantages and challenges,with current research focusing on enhancing efficiency, reducing energy consumption, and minimizing environmental footprint. The classical esterification of carboxylic acids with alcohols is one of the most utilized methods due to the accessibility of carboxylic acids and alcohols. This reaction, however, is limited by its equilibrium nature and typically requires a strong acid catalyst, such as sulfuric acid, to protonate the carboxyl group, thereby enhancing electrophilicity. Traditional approaches to drive this equilibrium toward ester formation involve high reaction temperatures and the continuous removal of water [6]. Recent advancements have introduced heterogeneous acid catalysts such assulfonated carbon, zeolites, and mesoporous silicas which provide advantages in terms of recyclability and reduced catalyst contamination [7]. Furthermore, studies on microwave and ultrasonic-assisted esterification have demonstrated enhanced reaction kinetics and increased yields under milder conditions, which supports a reduction in energy consumption while maintaining high reaction efficiencies. Thesephysical activation methods are gaining prominence in green chemistry frameworks, particularly for large-scale applications. Esterification via acid anhydrides with alcohols offers a more reactive pathway than carboxylic acid-alcohol esterification. The electrophilic nature of acid anhydrides permits reaction under milder conditions and often without the need for a strong acid catalyst [8]. Acetic anhydride is frequentlyutilized in both laboratory and industrial contexts due to its reactivity and lower cost.This reaction pathway also minimizes the water by-product, facilitating higher yields and purer ester products. However, the generation of acetic acid as a by-product necessitates downstream separation,increasing processing costs. To address these limitations, recent studies have explored theuse of biodegradable anhydrides and alternative green solvents to reduce the environmental impact of this synthesis route. Additionally, ionic liquids have shown promise as dual-function reaction media and catalysts, enhancing the sustainability of acid anhydride esterification in synthetic applications. The reaction of acid chlorides with alcohols represents one of the most efficient routes forester synthesis, characterized by rapid reaction kinetics due to the high electrophilicity of acid chlorides. This method, which does not require a catalyst, typically yields high-purity esters at ambient temperatures, making it favorable insynthetic applications where reaction speed and efficiency are critical [9]. However, the by-product hydrogen chloride gas poses challenges, such as equipment corrosion and the need for neutralization. To mitigate these issues, novel activating agents like N,N'-carbonyldiimidazole (CDI) and dicyclo-hexylcarbodiimide (DCC) have been investigated as substitutes for acid chlorides, enabling ester formation while reducing the environmental and safety concerns associated with hydrogen chloride [10]. Additionally, recent advancements in green chemistry have focused on replacing conventional acid chlorides with more benign electrophiles, thereby reducing toxic by-products while maintaining high reactivity. To align ester synthesis with green chemistry principles, recent research has explored altenative reaction media and catalytic systems that reduce or eliminate traditional organic solvents. Methods such as transesterification using renewable feedstocks (e.g., vegetableoils) have been employed to producebiodiesel and other biodegradable esters, providing an eco-friendly approach to estersynthesis. The application of deep eutectic solvents (DES) and supercritical fluids as reaction media has been shown to increase reaction efficiency while facilitating catalyst recovery and recycling [11]. These solvents exhibit unique physicochemical properties that support high selectivity and yield, making them suitable for large-scale ester production with minimized environmental impact. Additionally, the use of metal-organic frame-works (MOFs) and supported ionic liquid phases (SILPs) as catalytic materials is emerging as a promising strategy for enhancing catalyst reusability and optimizing reaction conditions.
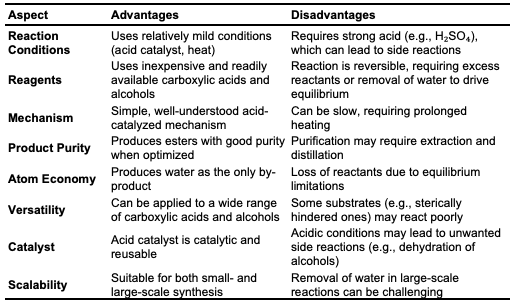
Fischer Esterification Fischer esterification is a traditional and widely used method for synthesizing esters, involving the reaction of carboxylic acids and alcohols in the presence of a catalyst under heated conditions (Figure 1). Typically, this process uses refluxing excess alcohol with organic acids, catalyzed by a concentrated mineral acid, at an elevated temperature for several hours [12].
Recent research has focused on optimizing Fischer esterification to enhance reaction efficiency, sustainability, and industrial applicability. Heterogeneous acid catalysts, such as sulfonated polymers and zeolites, have been developed as alternatives to traditional homogeneous acids like sulfuric acid. These catalysts offer higher catalytic efficiency, recyclability, and reduced environmental impact. For instance, studies [13] reported the use of sulfonated graphene oxide, resulting in improved yields and reusability. Additionally, ionic liquids and deep eutectic solvents (DES) have been reviewed as eco-friendly solvents that enhance esterification rates and minimize hazardous reagent use. Nonetheless, the process has some limitations, such as the high boiling point, slow reaction rate, and occasionally low yields [6]. Studies have shown that conventional ester synthesis typically requires between 1 to 4 hours for completion. Research revealed that synthesizing methyl esters requires over 1 hour to achieve complete conversion under standard reflux conditions [14].
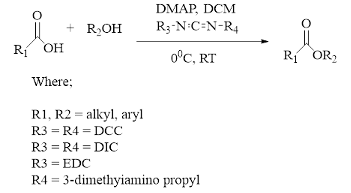
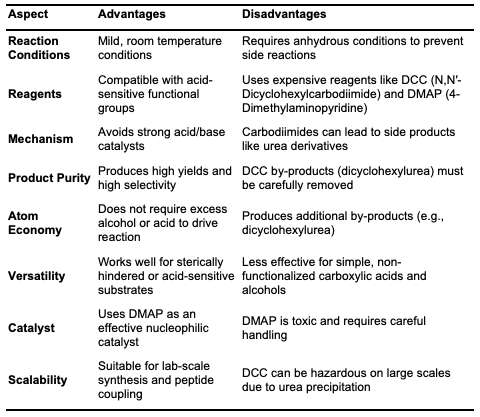
Steglich Esterification Steglich esterification involves the conversion of acids and alcohols into esters under neutral and mild conditions (Figure 2).
It is considered one of the most efficient methods for ester synthesis [15]. This reac- tion typically employs carbodiimide coupling reagents, such as N,N'-dicyclohexyl- carbodiimide (DCC), N,N'-diisopropyl-carbodiimide (DIC), and N-ethyl-N'-(3- dimethylaminopropyl)carbodiimide (EDC). For example, DCC reacts with a carboxylic acid in the presence of dimethylamino-pyridine (DMAP) to facilitate ester formation. The reaction mechanism involves the initial interaction between the carboxylic acid and carbodiimide to form an intermediate, known as O-acyl urea. The acyl group from this intermediate is then transferred by DMAP, which interacts with the alcohol to produce an ester. Here, DMAP acts as an activating agent to transfer the acyl group [16]. Steglich esterification has been used to synthesize several natural products, including anaenamides, cephalosporins, laterocidine, havellockate, and higginasianin [15].
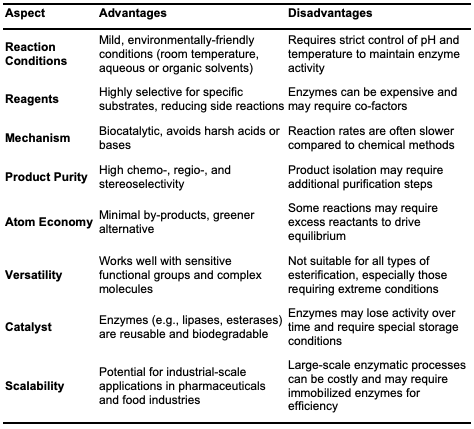
Enzymatic Esterification Enzymes are non-toxic biocatalysts that accelerate the rate of reactions [17]. They are used in reversible reactions and are highly versatile in the catalysis of various types of reaction using mild conditions. Enzymes have gained considerable attention in synthetic chemistry due to their versatility, high specificity, chemo-selectivity, stereo- selectivity, regio-selectivity, their broad substrate array as well as their high reaction yield ability in organic chemistry. Notably, enzymes can selectively catalyze the formation of esters at specific positions on a molecule and can produce a single enantiomer (chiral molecule) preferentially. Enzymes are environmentally friendly due to the use of mild conditions and biodegradability. They avoid the use of hazardous chemicals, reduce waste generation, and often allow for aqueous or solvent-free reactions, aligning with the principles of green chemistry [18]. As a result of enhanced regioselectivities, synthesis of sucrose esters gives a higher purity form. In enzymatic esterification, the enzyme binds to the substrates (the alcohol and the acid) at its active site, stabilizing the transition state and lowering activation energy to accelerate the reaction. The process typically proceeds through the formation of an acyl-enzyme intermediate, followed by the nucleophilic attack of the alcohol, leading to the release of the ester and regeneration of the enzyme. Enzymes as a catalyst have gained importance and significant use in the biotechnology sector globally today. Enzymatic esterify-cation has been experiencing a resurgence in interest and innovation due to its potential for more sustainable and efficient chemical synthesis [19]. The use of enzymes as a catalyst in the synthesis of esters are economically friendly. Although enzymes are expensive, their 7 recyclability and the reduced need for purification steps offset the costs. Enzymes are often reused multiple times without significant loss of activity, contributing to the overall cost-efficiency of the process [20]. Enzymatic esterification is one of the friendly and biological ways of producing biodiesel, compared to that of chemical means in term of temperature, reaction time, purification, desired product, recovery and lot more [21]. Several types of enzymes, including lipases, esterases, pro-teases, carboxylesterases, and phosphor-lipases, can promote ester formation. Among these, lipases are the most commonly used due to their ability to catalyze esterification and transesterification of a wide range of substrates, as well as their high stability, versatility, and relatively low cost [22]. Lipase, also known as triacylglycerol ester hydrolase, is a hydrolase enzyme widely utilized in organic synthesis and biotechnology due to its broad temperature and pH stability, high enantioselectivity, and substrate specificity [23]. Lipases catalyze the hydrolysis of lipids into fatty acids and glycerol and can facilitate reactions involving alcohols and carboxylic acids, esters and alcohols, esters and acids, and acyl group transfer from esters to nucleophiles. Although lipases are found across various organisms, industrial applications primarily utilize those derived from yeast and fungi. Lipases are instrumental in biodiesel production from waste cooking oil. In ester syn-thesis, lipase functions at equilibrium (Figure 3) and requires a small amount of water to maintain catalytic activity. When anhydrous organic solvents are used, product ester hydrolysis can occur, affecting yield [24].
Esterase is another enzyme used in esterification, recognized for its specificity and efficiency under mild conditions. As a member of the hydrolase enzyme class, esterases act on ester bonds and can operate in both esterification (forward) and hydrolysis (reverse) reactions. In esterase- catalyzed esterification, the enzyme catalyzes the formation of an ester from a carboxylic acid and an alcohol [25]. Unlike lipases, which hydrolyze long-chain, insoluble carboxylic acids, esterases typically hydrolyze short-chain acids and play a significant role in the metabolism of various biomolecules. Esterases act on substrates like fats, oils, and other ester-containing compounds. They are used in the synthesis of flavors, fragrances, pharmaceuticals, and biofuels [26]. The mechanism involves nucleophilic attack by the hydroxyl group of the alcohol on the carbonyl carbon of the acid, forming a tetrahedral intemediate that subsequently collapses to release water and form the ester bond. The enzyme stabilizes the transition state, lowering the activation energy and enhancing the reaction rate [27]. Protease, another enzyme involved in esterification, traditionally cleaves peptide bonds but can also catalyze ester bond formation under specific non-aqueous or low-water conditions, broadening its utility in organic synthesis and biotechnology [28]. This versatility makes proteases valuable in sythetic applications where traditional hydrolysis activity is minimized. Carboxylesterases, another hydrolase sub- class, hydrolyze carboxylic esters into alcohols and carboxylates and are essential for drug metabolism and detoxification. They play a significant role in pharmaceutical research, where they transform ester-containing drugs into active or inactive forms [29]. In addition, carboxylesterases are used in agrochemical detoxification, bioremediation, and the synthesis of fine chemicals and bio-fuels [30]. Their catalytic mechanism involves a nucleophilic attack on the carbonyl carbon of the ester, forming a tetrahedral intermediate and an acyl-enzyme complex. Subsequent hydrolysis of this complex by water releases the carboxylate product and regenerates the enzyme.
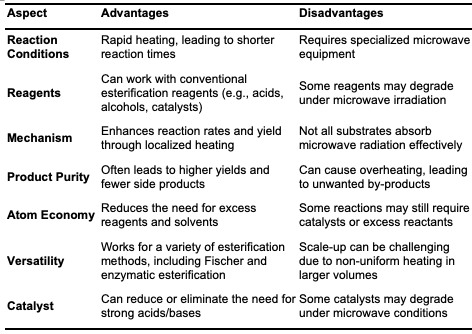
Microwave- Assisted Esterification Microwave-assisted esterification is a versatile and energy-efficient synthetic route for ester synthesis, offering advantages such as reduced reaction times, cost-effectiveness, and positive environmental impacts [31]. Microwaves are electromagnetic waves with frequencies ranging from 300 MHz to 300 GHz, used widely in organic synthesis. This technique relies on dipolar polarization and ionic conduction, with low energy input, allowing for efficient energy transfer through electromagnetic waves [32]. During microwave-assisted esterification, the reac- tion medium is irradiated, causing polar molecules, such as alcohol, to align with the oscillating magnetic field. This alignment, coupled with molecular dipole interactions with the electric field, generates heat through molecular friction, thereby accelerating reaction rates.
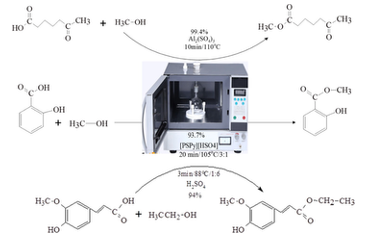
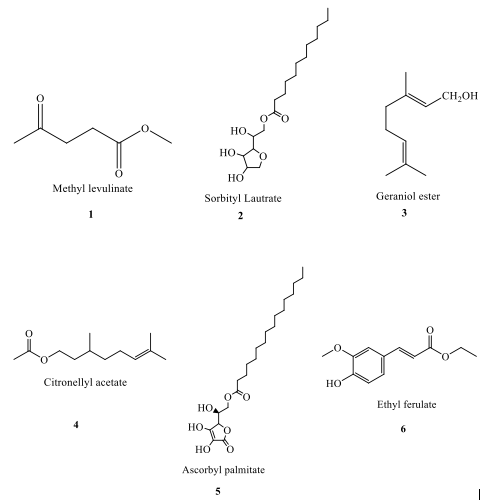
Microwave-assisted synthesis has proven effective for biodiesel production and has been successfully applied to the synthesis of esters like methyl levulinate, ethyl ferulate, and methyl salicylate, as shown in Figure 4. Esterification reactions using microwave irradiation can be conducted in various setups, including batch systems and continuous flow reactors [33].
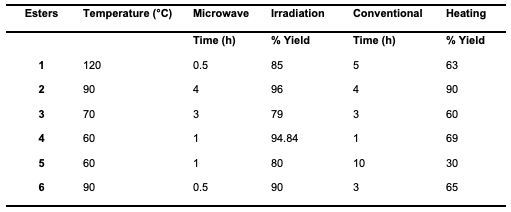
This technique addresses limitations associated with conventional esterification methods, making it a valuable approach for synthesizing ester compounds. Table 5, below, provides a comparative analysis of reaction times and yields for synthesizing various esters using microwave irradiation versus conventional heating methods, highlighting the efficiency and benefits of microwave-assisted synthesis in terms of both reaction speed and product yield.
Studies show that microwave irradiation significantly reduces the reaction time compared to conventional heating. [34] reported the synthesis of methyl levulinate (1) at 120°C, takes only 0.5 h with 85% yield using micro-wave irradiation, while conventional heating requires 5 h and yields only 63% [34]. This trend is similarly observed for ascorbyl palmitate (5), where microwave irradiation at 60°C yields 80% in just 1 h, whereas conventional heating requires 10 h and produces a much lower yield of 30% [35]. The effectiveness of microwave irradiation is further demonstrated in the synthesis of ethyl ferulate and citronellyl acetate. At 90°C, ethyl ferulate (6) synthesis takes only 0.5 h with microwave heating, yielding 90%, whereas it requires 3 h with conventional 12 heating and achieves a lower yield of 65% [6]. Similarly, for citronellyl acetate (4) at 60°C, microwave irradiation yields 94.84% in 1 h, compared to 69% yield with conventional heating over the same time period [36]. However, esters like sorbityl laurate and geraniol ester, the reaction times are similar across both methods, suggesting that micro-wave irradiation does not always drastically reduce reaction time. For sorbityl laurate (2), both methods require approximately 4 h, though microwave heating provides a slightly higher yield of 96% compared to 90% with conventional heating [37]. Also, the synthesis of geraniol ester (3) at 70°C takes 3 h with both methods, though the yield with micro-wave irradiation (79%) is higher than that of conventional heating (60%) [38].
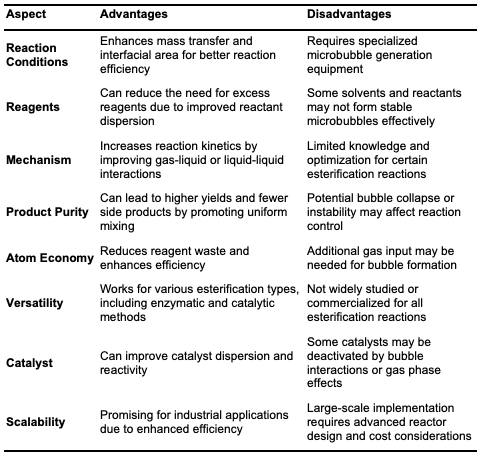
Microbubble Esterification Microbubble technology is an innovative method for enhancing esterification efficiency, particularly in biodiesel production from vegetable oils, as well as in synthesizing esters for pharmaceuticals and fragrances. Microbubbles, generated using a micro-bubble generator, have diameters between 10 and 100 micrometers [39]. This technique can be implemented through methods such as ultrasonics, gas sparging through porous materials, and mechanical agitation. Studies have shown that microbubbles significantly improve mass transfer at the gas-liquid interface, providing a stable surface area where reactants can easily diffuse to the reactive sites [6]. The small size and high surface area of microbubbles allow for uniform distribution and effective mixing within the liquid medium, optimizing reaction conditions. For example, reactions between carboxylic acids and alcohols, such as oleic acid with methanol or ethyl acetate production, demonstrate higher yields in shorter reaction times when using microbubble reactors [40].
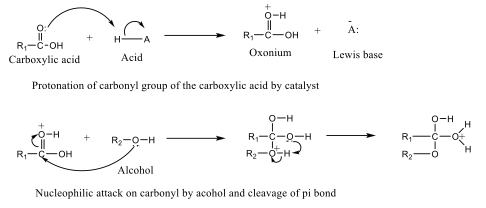
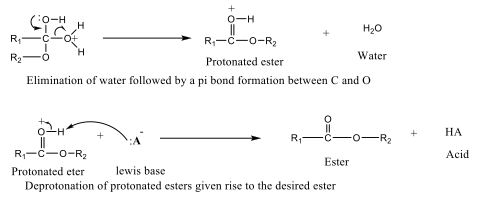
Acid- and Base-Catalyzed Esterification Esterification of alcohols with carboxylic acids in the presence of an acid catalyst is known as acid-catalyzed esterification. This process (Figure 5) involves the protonation of the carboxylic acid, which increases the electrophilicity of the carbonyl carbon, making it more susceptible to nucleophilic attack by alcohol.
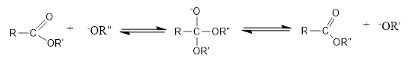
Following nucleophilic attack and subsequent proton transfer, the product undergoes dehydration to form the ester. Common acid catalysts include sulfuric acid (H₂SO₄) and hydrochloric acid (HCl), and the reaction mixture is typically heated under reflux to facilitate the reaction [41]. Base-catalyzed esterification refers to the reaction between an ester and an alcohol in the presence of a base catalyst to form a new ester (Figure 6). This reaction is widely used in the synthesis of fatty acid esters and in biodiesel production from natural products [42-44].
In this process, the base deprotonates the alcohol, generating an alkoxide ion, a strong nucleophile that attacks the carbonyl carbon of the ester. This forms a tetrahedral intermediate, which then undergoes further rearrangement to produce a new ester and an alkoxide ion. Base-catalyzed esterification generally proceeds faster than acid-catalyzed esterification [45]. However, one limitation of base-catalyzed transesterification is the risk of saponification, where fatty acids react with the base to form soap, leading to phase separation issues in the reaction mixture.
Reaction Mechanism of Esterification
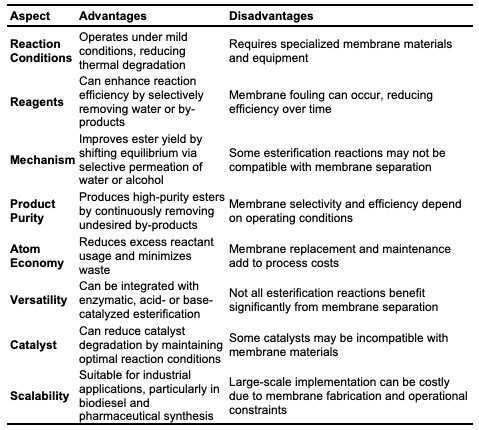
Membrane Technology Esterification Membrane technology has gained significant attention for its role in the synthesis and purification of esters. This technique utilizes selectively permeable membranes to enhance various stages in ester production, including separation, purification, and catalysis [46]. It has proven especially valuable in applications such as biodiesel synthesis. Membrane technology enables the continuous removal of by-products (e.g., water) from esterification reactions, shifting the reaction equilibrium toward product formation and increasing both yield and reaction rate. It also allows selective removal of impurities and unreacted starting materials, resulting in high-purity ester products. Membrane technology is suitable for both laboratory-scale synthesis and industrial production of esters, offering advantages in energy efficiency, sustainability, and cost-effectiveness. Various membrane technologies are used in ester synthesis, including pervaporation, membrane distillation, membrane bioreactors, nano-filtration, and catalytic membranes, each classified by mode of action and application in the esterification process. 17 Pervaporation, a widely used membrane technology in ester synthesis, selectively removes water from the esterification reaction mixture [47]. This method is especially useful for separating close-boiling-point mixtures and low-concentration compounds. In pervaporation, a hydrophilic membrane separates reactants from the permeate side, where water vapor is continuously removed. literature reported successful synthesis of ethyl acetate via the esterification of acetic acid and ethanol, using polyvinyl alcohol (PVA) and polyacetate membranes with high selectivity for water removal [48]. This selective removal of water shifted the equilibrium toward ester formation, achieving higher yields. Membrane distillation employs a hydrophobic membrane to separate water based on differences in vapor pressure and is frequently used in high-temperature reactions [49]. Research has demonstrated the application of polytetrafluoroethylene (PTFE) membranes in the esterification of lactic acid with methanol [50]. This study found that PTFE membranes offer stability and selectivity, making them suitable for continuous water removal at elevated temperatures, significantly enhancing both reaction rate and ester yield. Membrane bioreactors (MBRs) are commonly used in enzymatic esterification, combining enzymatic catalysis with membrane separation to provide controlled reaction environments and efficient product recovery [51]. Enzymes immobilized on the membrane surface offer a high surface area for reactions, while the membrane selectively removes water, resulting in high conversion rates and purity of the ester products. Nanofiltration membranes, characterized by nanometer-range pore sizes, are also frequently used in ester synthesis for their 18 ability to separate small molecules [52]. These membranes retain the ester product while allowing the passage of water and unreacted alcohol, which increases ester concentration and yield [53]. Catalytic membranes, incorporating catalytic sites within the membrane matrix, integrate reaction and separation into a single unit and have shown significant promise in ester synthesis. [54] reported the use of a sulfonated polyether ether ketone (SPEEK) catalytic membrane for the esterification of oleic acid with methanol. The SPEEK membrane provided catalytic sites for the reaction and allowed for continuous water removal, enhancing the overall efficiency and yield of the esterification process.
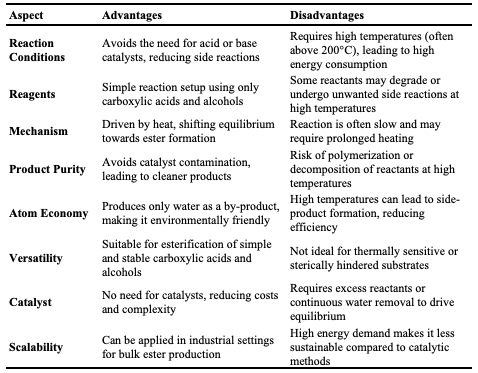
Non-Catalytic Thermal Esterification Non-catalytic thermal esterification is another synthetic route for ester production conducted without the use of catalysts, relying solely on thermal energy to facilitate the reaction between carboxylic acids and alcohols [55]. In this approach, heat provides the activation energy required for the reaction to proceed. Non-catalytic thermal esterification employs alcohol in either supercritical or subcritical states [56]. In the supercritical state, alcohols, such as methanol, are heated above their critical temperature and pressure, resulting in a fluid with properties between those of a liquid and a gas, which enhances solubility and reaction rates. Supercritical fluids promote efficient mixing, diffusion, and reaction kinetics, resulting in desirable yields. For example, [57] reported the synthesis of methyl oleate from oleic acid using supercritical methanol. Another study documented the production of fatty acid methyl esters (biodiesel) from waste beef tallow using supercritical methanol, demonstrating that the supercritical approach enables a green synthetic pathway with high conversion rates. At supercritical temperatures, methanol’s dielectric constant is reduced, forming a single-phase mixture that favors the esterification reaction. In the subcritical state, alcohol is heated to high temperatures without reaching its critical point, allowing it to act as a liquid or gas [58]. Subcritical fluids offer reduced solubility and diffusion rates, potentially resulting in slower reaction kinetics but with operational advantages. For instance, esterification of fatty acids with ethanol in subcritical water can produce ethyl esters [59]. Additionally, reported the synthesis of fatty acid methyl esters from fatty acids derived via subcritical water hydrolysis of fresh and waste cooking oil. This study 20 highlighted that subcritical water hydrolysis enhances esterification by increasing the ionic product, thus facilitating the reaction, and reducing the need for extensive feedstock purification. The mechanism of action in non-catalytic thermal esterification involves nucleophilic acyl substitution [6]. At elevated temperatures (150–250°C), the hydroxyl group (-OH) from the carboxylic acid and the hydrogen atom (H) from the alcohol combine to form water (H₂O). The continuous removal of water from the reaction shifts the equilibrium toward ester formation. Non-catalytic thermal esterification offers several advantages, including the elimination of catalyst-related costs, reduced contamination, and fewer purification steps. These benefits are particularly valuable in biodiesel and fragrance production, where catalyst residues can complicate product quality. However, this method also has limitations, such as the need for high temperatures (often above 200°C), which increases energy consumption and can limit reaction rates, particularly with bulky or less reactive substrates [6].
Factors Affecting the Synthesis of Esterification Esterification is a widely studied organic synthesis process due to its applications in pharmaceuticals, industry, cosmetics, food, beverages, and personal care products. However, several factors impact the reaction chemistry of esterification. The mode of reaction during synthesis significantly affects esterification. According to [6], esterification involves the equimolar reaction of a carboxylic acid with an alcohol. However, the equilibrium constant for this reaction is influenced by both temperature and the ratio of reactants. Typically, only two-thirds of the acid reacts in an equimolar mixture; thus, increasing the alcohol concentration enhances product yield. However, an excess of alcohol can reduce the catalyst concentration, which may hinder the forward reaction and lead to catalyst deactivation due to binding at the active site [60]. The structure of the reactants also affects esterification chemistry (Figure 7). Alcohols are classified as primary, secondary, or tertiary, based on the position of the hydroxyl group.
This classification impacts ester yield and by-product formation. Primary alcohols are reported to yield high ester levels within a short reaction time, secondary alcohols yield an average ester percentage, and tertiary alcohols tend to produce a low yield and require a longer reaction time due to steric hindrance around the hydroxyl group [6]. Water is another critical factor influencing esterification chemistry. Water shifts the reaction equilibrium toward the reactants, which can reduce ester yield. However, continuous removal of water from the reaction mixture promotes the forward reaction, resulting in a higher ester yield. Water presence can also lead to ester hydrolysis, impacting the reaction rate. Acid-catalyzed esterification is particularly sensitive to water, as it can lower catalytic activity and slow down the reaction rate [61]. Catalysts play a vital role in esterification by lowering activation energy, increasing reaction rates, and influencing reaction conditions [62]. The choice of catalyst affects process efficiency, selectivity, and sustainability. Solvents also impact reaction rates; excessive solvent use, especially polar or hydrophilic solvents, can reduce ester yield. Polar solvents may interact with the enzyme's water layer, altering its activity, whereas non-polar solvents enhance enzyme dispersion at active sites, resulting in a higher reaction rate and yield [63]. Additionally, polar solvents can stabilize ionic intermediates and transition states, which can benefit the reaction rate. In acid-catalyzed esterification, polar solvents dissolve the carboxylic acid, alcohol, and catalyst, facilitating the reaction. However, an excess of polar solvent may slow down the reaction.
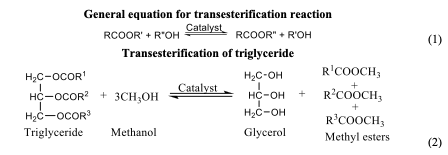
Transesterification Transesterification is a reaction method that involves the conversion of oils and fatty acids into alkyl esters. The process involves a reaction of triglycerides with an alcohol (alkane) to form alkyl ester of fatty acids and glycerol. It is a versatile reaction that is widely used in the production of biodiesel, polymers, and fine chemicals [64]. Transesterification, also known as alcoholysis, entails the reaction of lipids and low carbon alcohol in the presence of a homogeneous or heterogeneous catalyst [65]. It is also used in the production of hardened vegetable oil as it reduced its viscosity and improved its fuel properties. Methanol and ethanol are usually employed in the transesterification process, but methanol is preferred because of it is economically friendly nature and steady availability. As many underutilized seed oils are emerging, the transesterification to fatty acid methyl or ethyl esters prior to analysis have been widely adopted [66,67]. The application of the transesterification techniques extended to analysis of triacylglycerol (fats and oils) from animal sources [68]. Transesterification process can be carried out through homogenous or heterogeneous acid or base catalysts. Generally, it is believed that homogeneous catalysis undergoes a fast reaction when compared to the heterogenous catalyst; yet, it cannot be reused, which serves as a major setback. However, the use of heterogenous catalysis in continuous biodiesel production results in high activity, high selectivity, ease of separation from product and catalyst reusability, although its reaction often takes longer [69]. Despite the time taken for the reaction to complete, heterogeneous plays a major role in overcoming the challenges faced by homogeneous catalysis. This includes the ability to regenerate and recycle the products, quick separation and purification of product. There are some factors affecting the transesterification process which include the type of catalyst used and concentration, mole ration, reaction time, temperature, and free fatty acid [70].
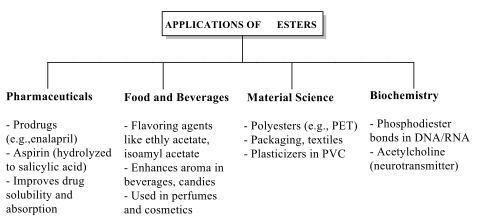
Application of Esters Ester compounds are one of the major compounds used across different industries due to their distinctive chemical properties. Synthetic esters are extensively used as intermediates in the formation of a variety of organic compounds. Literature revealed its application in the pharmaceutical industry, food and beverage industries, biochemistry, material sciences and so on. Reactive capacity of esters, particularly in esterification and hydrolysis reactions, makes them invaluable in the synthesis of pharmaceuticals, where they are used to create prodrugs [71]. These prodrugs improve the solubility of active pharmaceutical ingredients. Esters enhance the pharmacokinetic properties of active drugs. An example is the ester enalapril, a prodrug that is hydrolyzed in vivo, then converts to the active form of enalaprilat, used to treat hypertension and heart failure. Also, aspirin (acetylsalicylic acid), an ester that undergoes hydrolysis to produce salicylic acid, provides both anti-inflammatory and analgesic effects when used. These ester modifications improve solubility, stability, and absorption of drugs, enhancing their therapeutic efficacy [72]. In the food and beverage industry, according to [73], ester compounds are widely used as flavoring agents due to their ability to impart fruity and floral aromas. Different ester compounds such as ethyl acetate, isoamyl acetate, ethyl butyrate, methyl butyrate were studied. They impart fruity and floral scents to enhance the aroma and taste of a wide 24 range of products. Natural and synthetic esters are added to candies, beverages, and baked goods to achieve desired flavor profiles. In fragrances, esters are major components in the formulation of perfumes and cosmetics, contributing to the pleasant scents [74]. Esters have also been found useful in biochemistry, especially in biological processes for the formation of essential biomolecules. Esters are found in DNA and RNA as phosphodiester bonds, linking nucleotides together to form the backbone of genetic materials [75]. Literature has revealed acetylcholine, a neurotransmitter used in muscle contraction and neural signaling [76]. Ester compounds have also been widely used in material science, particularly in the production of polymers. Polyesters, such as polyethylene terephthalate (PET), are synthesized through the polycondensation of diols and dicarboxylic acids or their ester derivatives. These materials are used in the production of textiles, packaging, and plastic bottles due to their durability and flexibility [77]. Additionally, esters serve as plasticizers in the manufacturing of plastics, enhancing the flexibility and workability of polymers like polyvinyl chloride (PVC) [78].
Esters Toxicity Esters are widely used across industries, including food flavoring, fragrances, pharmaceuticals, and as solvents, making it essential to understand their toxicity profiles. Generally, esters exhibit low toxicity compared to other organic compounds, which makes them relatively safe for many applications. However, toxicity can vary significantly depending on ester structure and functional groups. Common esters, like ethyl acetate and methyl acetate, are low in toxicity and are frequently used in the food and beverage industries without significant health concerns when regulated appropriately [79]. However, the metabolic breakdown of esters can produce potentially toxic metabolites. For example, methanol (a breakdown product of methyl esters) is highly toxic and can lead to serious health effects, including blindness and central nervous system damage. Additionally, aromatic esters such as benzyl acetate can metabolize into benzyl alcohol and acetic acid, which may exhibit toxicity 25 depending on exposure dose and duration [80]. Long-term exposure to certain phthalate esters, widely used as plasticizers, is linked to adverse health effects such as endocrine disruption, reproductive toxicity, and developmental toxicity in humans and animals. Phthalates like diethyl phthalate disrupt hormone functions, posing risks to reproductive health and development, especially in infants, following prolonged exposure [81]. Regulatory bodies are working to limit the use of certain phthalates in consumer products, especially those intended for children and pregnant women, to reduce potential risks. In industries, exposure to high concentrations of ester vapors may lead to respiratory irritation, dizziness, headaches, and, in severe cases, central nervous system depression [82]. Thus, implementing protective measures, like proper ventilation and personal protective equipment, is essential to minimize these risks. Many esters are biodegradable and environmentally low risk, but breakdown products like certain phthalates can accumulate and impact aquatic ecosystems [83]. In response to concerns about ester toxicity, several studies have proposed solutions to reduce toxicity by modifying ester structure, controlling exposure, enhancing biodegradeability, and strengthening regulatory frameworks. Structural modifications can enhance compound safety by focusing on esters that yield non-toxic metabolites. [84] reported that switching from methyl to ethyl or personal protective equipment, is essential to minimize these risks. Many esters are biodegradable and environmentally low risk, but breakdown products like certain phthalates can accumulate and impact aquatic ecosystems [83]. In response to concerns about ester toxicity, several studies have proposed solutions to reduce toxicity by modifying ester structure, controlling exposure, enhancing biodegradeability, and strengthening regulatory frameworks. Structural modifications can enhance compound safety by focusing on esters that yield non-toxic metabolites. [84] reported that switching from methyl to ethyl or isopropyl esters in various applications reduces toxic metabolite formation, particularly avoiding methanol release, which has neurotoxic effects. Additionally, research shows that improving ester biodegradability has led to the development of alternative plasticizers, such as citrate and adipate esters, which are less toxic and more environmentally friendly than phthalates, decomposing readily without harmful byproducts [85]. Advanced ventilation systems and the use of PPE effectively reduce inhalation risks for workers exposed to volatile ester compounds, as high concentrations of ester vapors can cause respiratory and neurological issues.
Conclusion This study provides a comprehensive overview of esterification, synthesis methods, influencing factors, applications, and toxicity of esters. Esterification, a fundamental reaction in organic chemistry, plays a critical role in the synthesis of esters, which are widely utilized across industrial, pharmaceutical, and biochemical sectors. Esters are synthesized through the reaction of carboxylic acids and alcohols in the presence of catalysts, highlighting the versatility and efficiency of this chemical process. Advanced methods, including enzyme-catalyzed esterification and the use of alternative energy sources like microwave and ultrasonic irradiation, have further broadened the scope of ester synthesis, offering environmentally-friendly and efficient pathways. Several factors influence esterification, such as the nature of the reactants, catalyst type and concentration, temperature, and solvent choice. Understanding these factors is essential for optimizing reaction conditions to achieve high yields and selectivity. Esters have a wide range of applications across various sectors. In the pharmaceutical industry, esters are used as intermediates in the synthesis of drugs and prodrugs. The flavor and fragrance industries rely heavily on esters for their pleasant aromas and flavors. Additionally, esters serve as solvents, plasticizers, and lubricants in industrial applications, underscoring their multifunctional nature. The toxicity of esters is an important consideration in pharmaceutical and environmental applications. While many esters are generally regarded as safe, some can exhibit toxicity, necessitating thorough evaluation and regulation. Understanding the toxicological profiles of esters is crucial for mitigating potential risks and ensuring safe usage. Research in esterification and ester synthesis is a dynamic and evolving field, driven by the ongoing pursuit of more efficient, sustainable, and environmentally-friendly synthetic methods. The diverse applications of esters, alongside an awareness of their toxicity, enhance the significance of this class of compounds in modern science and industry. Future research and innovation in ester chemistry will undoubtedly lead to new discoveries and advancements.
References
|
|||||||||||||||||||||||||||||||||||||||||||||||||||