|
Introduction Natural organic matters (NOM), which are a mixtures of decomposition product of plants and animals, are major constituents commonly present in water that require removal [1,2]. Humic acid (HA) accounts for a significant portion (40-90%) of the NOM [3] and has a harmful effect on the water quality, such as undesirable color (yellowish to brownish) and taste; serves as food for bacterial growth; binds with heavy metals and other organic substances to yield high concentrations of these substances and enhance their transportation in the water; reacts with chlorine during water treatment leading to the evolution of chlorinated organic compounds, some of them know as human carcinogens (trihalomethanes) [3-7]. As a result, humic acid is considered as impurities and needs to be measured for water studies. In the past years, several methods have been employed for removal of humic acid from aqueous solution. adsorption is one of the most-effective methods in removal of humic acid. Several kinds of adsorbents have been employed for humic acid adsorption includ- ing zeolite [8], polypyrrole-coated granules [9], chitosan [10-13], clay [14], activated carbon [15-17], and resins [18]. However, some of the previously mentioned adsorbent compounds have some shortcomings; for example, activated carbon is expensive and requires high operating cost and require fre- quent regeneration [3]. In recent years, many efforts have been focused on low-cost and abundant natural minerals sorbents for water and wastewater treatment adsorbents. The conventional filtration process, which uses sand as the filter media, has been found to be ineffective in removing humic substances. In water filtration, there has been a considerable interest in the use of granular media that can enhance the removal of humic substances [19]. One option is to replace the sand with modified polyolefin as the filter media. Polyolefin’s and polypropylene have a very low surface charge due to the non-polarity nature of the polymer and thus because the surface tension of these polymers ranges between 30 and 40 dynes/cm, the polymers would not attract any polar chemicals and hence the removal of these compounds would not be achieved. The polarity can be changed of this family of polymers by ionizing the surface. However, this is not a permanent surface change as it tends to reverse with time. For this reason, natural materials such as natural fibers (palm date leaf) and seaweed (Posidonia) were chosen in this study to be melt mixed with polypropylene and investigate the humic acid removal by the modified polypropylene. To date, no study related to adsorption of humic acid by natural fiber-modified polypropylene has appeared in the literature. The aims of this study are (1) to determine the ability of modified polypropylene to adsorb humic acid from aqueous solutions and (2) to examine the effect of different parameters such as contact time, adsorbate concentration, adsorbent weight and initial pH value on the adsorption process. It is hoped that this modest effort may substitute the deficiency in the literature on the modification of polypropylene using natural fibers materials such as palm date leaf and seaweed.
Materials and Methods Materials A commercially available polypropylene in the form of granular pellets from LUKOIL and used as the matrix [(Polypropylene Buplen 6631), melt index = 1-3 g/10 min (230°C, 2.16 Kg), density of 898-905 kg/m3]. The natural fibers (palm date leaf) and seaweed (Posidonia) were provided from the Libyan Polymer Research Center, while humic acid was purchased from agricultural equipment market.Preparation of Modified Polypropylene The method of production that was chosen for the modified polymer was extrusion as this method could lead to a large production volume and the physical size could be maintained relatively easily. Dried polypropylene pellets were melted, mixing once with palm date leaf fibers and once with seaweed fiber powder by using HAAKE mini twin-screw extruder CTW, with average screw speed of 35 rpm, and barrel temperature of 190°C. Blend samples of different percentages (1, 3 and 5 w%) were prepared and then were grinded and prepared for analysis and adsorption experiments. Characterization Techniques The contact angle measurements were carried out using ramè-hart instrument co. Model 200-F4 at room temperature. Two µL volume drops of water were deposited on the surface of the samples using a syringe. Pictures of the water drops were acquired through a digital camera positioned on a static contact angle analyzer. The θ of the contact angle was measured automatically from the image setup. Each contact angle value is an average of five measurements. Unmodified and modified forms of the polypropylene were characterized using Fourier-transformed infra-red spectroscopy (630 FTIR, Agilent). Measurements were taken in transmission mode at the range of 4000-580 cm-1 with resolution of 4 cm-1. Batch Adsorption Experiments Humic acid solution was prepared by dissolving a certain amount of humic acid in a known volume of deionized water. The solution was stirred for 2 h and filtered through a Whatman membrane filter (0.45 µm) before conducting the adsorption tests. A series of batch adsorption experiments were conducted to examine the adsorption isotherm, and the effect of solution pH values on the adsorption behaviors. In the adsorption experiments, a number of 100-ml flasks contained 20 ml of the humic acid solution with an initial pH value of 6 and an initial humic acid concentration in the range of 10-100 mg/l, respectively, were used. A 0.2-gram amount of the modified polypropylene was added to each of the flasks and the contents were stirred on a shaker at 120 rpm and at room temperature (25°C) for 1 h for adsorption to take place and reach equilibrium. The initial and final humic acid concentration in the solutions in each of the flasks was determined with an UV-visible spectrometer (Photolab 7600 UV-vis, Xylem Analytics, Germany) at 254 nm in 1 cm quartz cell. All experiments were performed in triplicate and average values were used for calculations. The percentage of HA removal (%) was calculated from initial HA concentration (C0, mg/L), and final HA concentration (Ce, mg/L) according to equation 1: The amount of humic acid adsorbed on the modified polypropylene at adsorption equilibrium, qe (mg/g) was calculated from the equation 2: where C0 and Ceare the initial and equilibrium humic concentrations, respectively (mg/L), V is the total volume of the suspension (L), and m the adsorbent mass (g). Adsorption Isotherms The adsorption capacity is the value that gives information about the feasibility of the adsorbents for removing the humic acid as a pollutant from water. The two classical models of Langmuir and Freundlich were tested to describe the adsorption equilibrium of HA onto both modified polypropylene with 1% palm leaf fibers and modified polypropylene with 1% seaweed. The Langmuir mode in the linearized form can be written as follows [20]: where qe is the amount of humic acid adsorbed per unit weight of the modified polypropylene granules at equilibrium concentration (mg g-1), Ce is the final concentration in the solution (mg l-1), qmax is the maximum adsorption at monolayer coverage (mg g-1), and Kl is the adsorption equilibrium constant (l mg-1) . The experimental isotherm data are also modeled with log-linearized Freundlich model in the format [20]: were qe and Ce have the same definitions as in equation 3, Kf is a Freundlich constant representing the adsorption capacity (mg g) (l g-1) n, and n is a constant depicting the adsorption intensity (dimensionless).
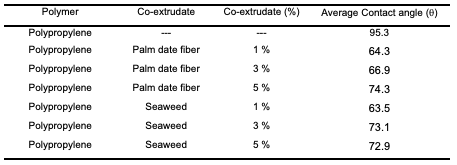
Results and Discussion Characterization of Modified Polypropylene Surface polarity The surface polarity or the repulsion of water was determined by means of the water droplet method and subsequent measurement of the contact angle of the water to the substrate. A one microliter droplet of distilled water was placed on the surface of the substrate to be measured. A photo was then taken with a stereomicroscope that was connected to a computer. The contact angle of the water droplet with the surface of the substrate was thus measured and reported as angle theta (θ). Data presented in Table 1 states that the presence of palm date leaf fiber and seaweed vastly reduces the surface tension, thus increases the polarity of the polypropylene. Furthermore, the lowest average contact angle or the highest polar surface was achieved with 1% seaweed followed by 1% palm date leaf fiber. Therefore, these two composites were used in the adsorption experiments.
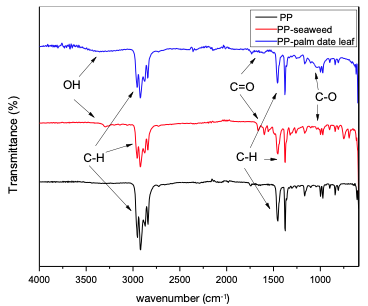
FTIR To obtain a clear understanding of the polypropylene structure characteristics, and the effects of natural fibers modifications, FTIR analysis was carried out. The obtained FTIR spectrum is shown in Figure 1.
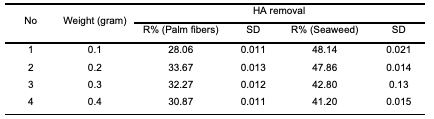
Figure 1 shows the FTIR spectrum of poly-propylene, before modification (original) and after modification with a 1% of palm dates leaf fibers, and with a 1% of seaweed. From Figure 1, FTIR spectra of polypropylene showed characteristic bands of polypropylene, stretching vibration of –C-H at 2959-2823 and bending vibrations of –CH2 and CH3 at 1471-1417 cm-1 and 1383- 1349 cm-1. When polypropylene was mixed with palm date leaf fibers and seaweed, new bands appeared that are characteristics of natural fiber and correspond to cellulose and lignin. O-H stretching vibration at 3448- 3169 cm-1 and carbonyl stretching C=O and C-O at 1770-1567 cm-1 and 1084-1043 cm-1, respectively. Adsorption of Humic Acid into Modified Polypropylene Effect of adsorbent weight on humic acid removal To investigate the effect of the modified polypropylene as an adsorbent on the efficiency of the humic acid adsorption process, the experiments were carried out with different weights of modified polypropylene ranging from 0.1, 0.2, 0.3 and 0.4 g; the other conditions were fixed for the adsorption process (initial humic acid concentration 20 mg/L, solution pH 2, temperature 25°C, and adsorption time 60 min, 120 rpm). Results are presented in Table 2.
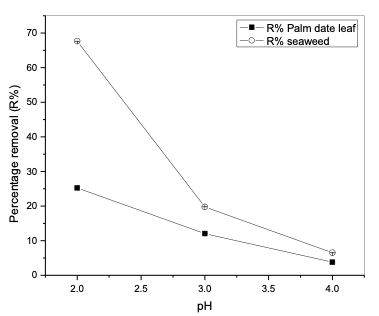
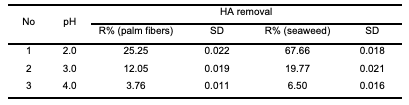
As was expected from the literature, when the adsorbent’s weight increased, the humic acid removal increased [21,22], but the results showed that the increase was not clearly noticeable and there was a fluctuation in the readings, especially in the case of the seaweed samples. However, the highest humic acid removal on both modified polypropylene samples was achieved when a weight of 0.2 g/20 mL was used at 25°C. From the results, it is clear that an increase in the adsorbent weight above 0.2 g had no important effect on the adsorption efficiency of humic acid from the aqueous solution. Additionally, regarding the efficiencies of two modifications, it is clearly noticed that more efficient removal of humic acid was achieved with polypropylene modified with seaweed than with polypropylene modified with palm date leaf fibers. Effect of pH on humic acid removal The effect of pH on the adsorption of humic acid onto modified polypropylene was studied by varying the pH of the solution from 2.0 to 5. The results are shown in Table 3 and Figure 2.
It was observed that the maximum adsorp- tion was achieved at pH 2.0. As the pH of the solution was increased from 2.0 to 4.0, the percentage removal of humic acid on modified polypropylene with palm date leaf fibers decreased from 25.25 to 3.76% at an initial concentration of 20 mg/L, while in the case of modified polypropylene with seaweed the humic acid percentage removal decreased from 67.77% at pH 2.0 to 6.5% at pH 4.0. On the other hand, results showed that there was almost no reduction in humic acid removal above pH 4.0. This can be explained by the following: as pH increases, both the adsorbent surface and the humic acid lose their protons and become negatively charged, and, consequently, repel each other. The decrease in humic acid adsorption with increasing pH suggests that electrostatic interaction plays a role in the adsorption process [23].
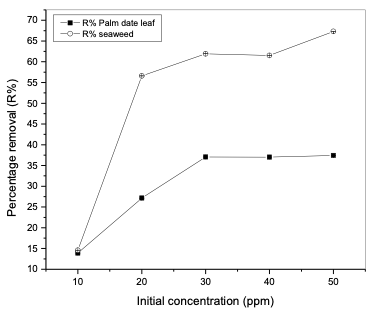
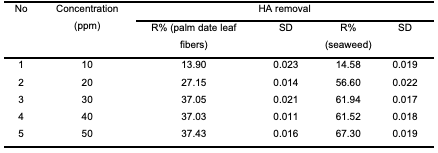
The high adsorption of HA at lower pH values can be attributed to the external hydrogen bonds formed between phenolic hydroxyl groups of HA and hydrogen bonding sites on the modified polypropylene [24]. The large increase in adsorption between pH 3 and 2 could also be attributed to a reduction in the size of the humic acid molecules at low pH [25]. Smaller molecular size enables more molecules to be adsorbed on the adsorbent’s surface. Effect of initial humic acid concentration In order to investigate the effect of initial humic acid concentration, experiments were carried out at room temperature (25°C) and at the initial pH 2.0. Initial humic acid concentrations ranging from 10 to 50 mg/L were prepared; 0.2 g of modified polypropylene was added to 20 ml of humic acid solution and agitated magnetically at 120 rpm for 60 min. Obtained results of humic acid removal are shown in Table 4 and Figure 3.
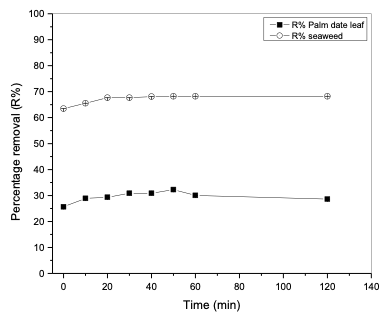
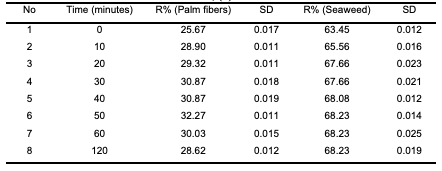
It can be observed that as the concentration increased from 10 to 50 mg/L, humic acid removal percentage increased from 13.9 to 37.43% in the case of modified poly-propylene with palm date leaf fibers, while in the case of modified polypropylene with seaweed, humic acid removal percentage increased from 14.58 to 67.3%. This is attributed to the increase in the mass driving force which allows more humic acid molecules to pass from the solution to the surface of the modified polypropylene. It is clear that the initial humic acid concentration plays an important role in the adsorption process. Effect of contact time The effect of agitation time on the ad- sorption of humic acid onto modified polypropylene was determined within the following contact time intervals: 5, 10, 20, 30, 40, 50, 60 and 120 minutes. Experiments were performed at room temperature (25°C), at a fixed modified polypropylene weight of 0.2 g, pH 2 and initial humic acid con- centration of 20 mg/L. Samples were stirred for 120 minutes. The results of humic acid removal as a function of time are presented in Table 5 and Figure 4. The results showed that the time of 50 minutes was sufficient to achieve the maximum adsorption for both modified polypropylene types. As can be seen in the Figure 4, the adsorption process can be divided into two phases: the humic acid removal was initially rapid (fast) and then slowed down until it reached equilibrium at around 50 min for polypropylene modified with palm date leaf fibers and polypropylene modified with seaweed.
This can be attributed to the fact that in the beginning, a large number of vacant surface sites are available for humic acid. After a while, the remaining vacant surface sites are hardly occupied due to repulsive forces between the humic acid molecules in the aqueous solution and those on the adsorbent surface [26-28].
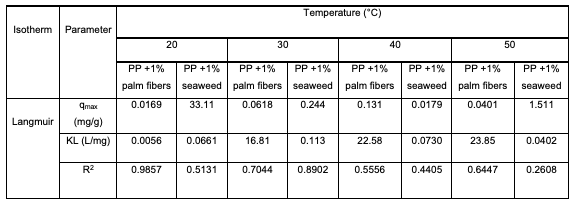
Adsorption Isotherm Models Two non-linear adsorption isotherm models were used to describe the humic acid adsorption onto tested modified polypropylene: Langmuir and Freundlich. The equilibrium adsorption data were processed by adsorption equations at temperatures of 20°C, 30°C, 40°C and 50°C, and the obtained adsorption parameters for humic acid adsorption onto two modified polypropylene adsorbents are presented in Table 6. The results show that the Langmuir isotherm model represents somehow well the experimental sorption data, since similar range of correlation coefficients (r2) were obtained. Namely, the r2 values for the Langmuir adsorption model were in the range from 0.513 to 0.986 at 20°C, and from 0.704 to 0.890 at 30°C. The calculated isotherm parameters revealed that HA adsorption onto polypropylene modifications (palm date leaf fiber and seaweed) followed the order: Langmuir. Therefore, HA adsorption onto polypropylene modified with (a) palm date leaf fiber and (b) seaweed can be characterized as monolayer adsorption.
It can be seen from Table 6 that the Langmuir maximum adsorption capacity qmax was found to be 33.11 mg/g for polypropylene modified with 1% seaweed at 20°C. On the other hand, the modified polypropylene with 1% palm date leaf fiber prepared in this work showed a low adsorption capacity of 0.0169 mg/g at the same conditions.
Conclusion While polypropylene has not been studied for humic acid removal due to its polarity, it is possible to modify its surface with some local natural materials to extend the application of polypropylene as an adsorbent to remove charged organic compound such as humic acid from water and wastewater. In this study, the removal of humic acid from aqueous solution using commercially available polyolefin (polypropylene) modified with natural fibers materials (palm date leaf fiber and seaweed) was investigated. The following conclusions are extracted from this study:
References
|
||||||||||||||||||||||||||||||||||||||